In recent years, global environmental concerns have prompted a shift toward eco-friendly manufacturing in the field of organic synthetic chemistry. In this regard, research into photoredox catalytic reactions, which use light to initiate redox or reduction-oxidation reactions via a photoredox catalyst, has gained significant attention. This approach reduces the reliance on harsh and toxic reagents and uses visible light, a clean energy source.
A key research area has been the development of recycling methods for photocatalysts, which offer both economic and environmental benefits. Photocatalysts use light to accelerate a chemical reaction without getting consumed in the process, and photoredox catalysts are photocatalysts specifically designed for redox reactions.

While recycling methods for heterogeneous photocatalysts, such as semiconductors and polymers, have been widely developed, there has been less focus on recycling organic photocatalysts. Considering the cost-effectiveness and low toxicity of organic photoredox catalysts, developing suitable recycling approaches is essential for achieving sustainable organic synthesis.
Addressing this gap, a team of researchers from Okayama University, Japan, including Assistant Professor Kenta Tanaka from the Research Institute for Interdisciplinary Science, along with then-graduate student Haru Ando, Associate Professor Hiroyoshi Takamura, and Professor Isao Kadota from the Department of Chemistry at the Graduate School of Natural Science and Technology, developed a novel phenothiazine-based organic photoredox catalyst. Their study was published in the journal Chemical Communications on March 19, 2024.
“Phenothiazines, or PTHs, are widely used as photocatalysts in organic chemistry,” explains Prof. Tanaka. “However, the high reactivity of the p-position relative to the nitrogen atom on 10-aryl phenothiazine molecules, makes them prone to reacting with electrophiles, reducing their stability.
“The development of more stable and sustainable photocatalysts is therefore highly desirable. To address this, we developed new phenothiazine-based photocatalysts that is both stable and recyclable.”
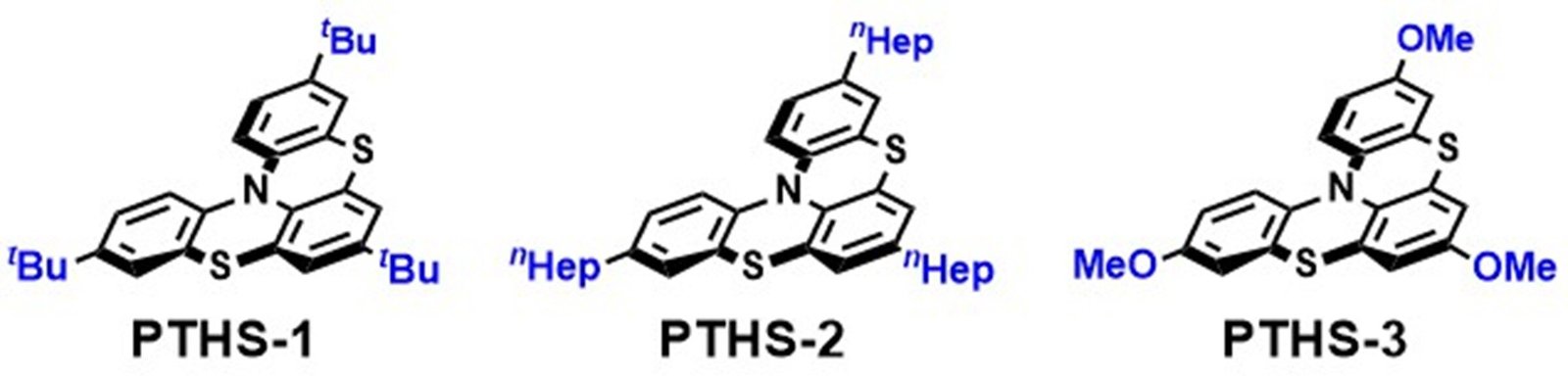
Their novel phenothiazine catalyst, called PTHS, features a spiral structure with a bulky electron donating group, called tBu, substituted at the p-position of the nitrogen atom, providing enhanced stability. The researchers developed a series of phenothiazine photocatalysts (PTHS 1–3) and evaluated their structural and physical properties via electrochemical and spectroscopic experiments. They discovered that the new catalysts possess a strong reducing ability and can be activated using blue light.
To test their stability, the team compared the new catalysts with existing PTH catalysts by subjecting them to photochemical sulfonylation reactions. The results revealed that while PTH could not be recovered and monosulfonylated product was obtained in 78%, 95% of PTHS could be recovered, indicating greater stability.
Additionally, the researchers tested the recyclability of the catalysts in a photochemical phosphonation and found that the catalytic activity of PTH, and thus reaction yield, decreased with repeated use. In contrast, PTHS-1 could be effectively recovered multiple times without any loss of catalytic activity and yield. Moreover, PTHS-1 is also suitable for large-scale synthesis, achieving 96% recovery even at gram-scale synthesis.
“The new phenothiazine photocatalysts have the potential to be applied to various visible-light-induced photochemical reactions, which was not possible with any of the hitherto reported phenothiazine photocatalysts. We believe our recyclable organic photocatalysts will be a promising tool for the efficient synthesis of various pharmaceuticals and functional materials,” remarks Ando.
Overall, these innovative photocatalysts represent a significant step for achieving sustainable organic synthesis, paving the way towards environment-friendly chemical manufacturing.
More information:
Haru Ando et al, Strongly reducing helical phenothiazines as recyclable organophotoredox catalysts, Chemical Communications (2024). DOI: 10.1039/D4CC00904E
Provided by
Okayama University
Citation:
Researchers develop organic photoredox catalysts with enhanced stability and recyclability (2024, May 24)
retrieved 24 May 2024
from https://phys.org/news/2024-05-photoredox-catalysts-stability-recyclability.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.