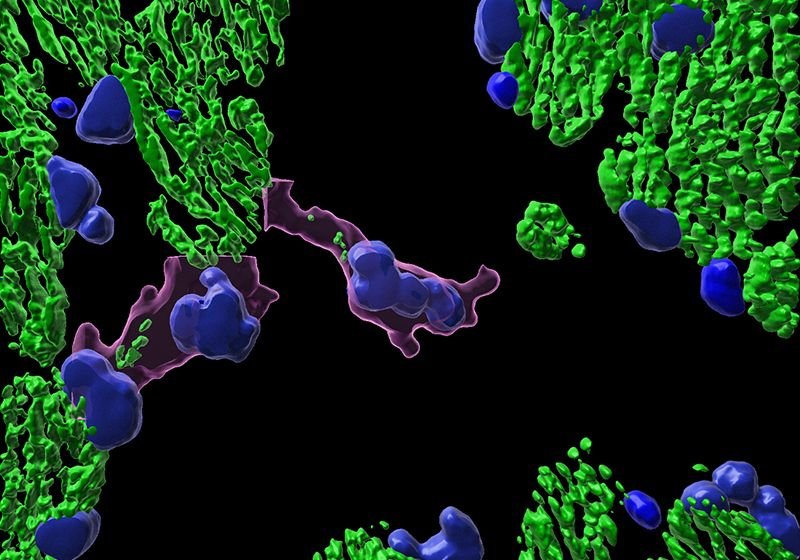
Organelle health is vital to a cell’s function. Consequently, cells have many mechanisms to repair or eliminate defective organelles. Åsa Gustafsson, a cellular and molecular biologist at the University of California, San Diego studies mitochondrial function and its importance to the cells of the heart. In a recently published Nature Communications paper, Gustafsson’s laboratory determined that cardiac myocytes and other cells use secretion to remove mitochondria from the cell when lysosomal degradation is inhibited.1 To investigate this process, Gustafsson and her team employed several microscopy techniques, including spinning-disk confocal microscopy, fluorescence microscopy, transmission electron microscopy, and correlated light and electron microscopy (CLEM).
What motivated you to perform this study?
Åsa Gustafsson from the University of California, San Diego investigates the pathways cardiac myocytes use to eliminate dysfunctional and damaged mitochondria.
Shakti Sagar

Mitochondria generate most of the cell’s energy. However, when they become dysfunctional, damaged, or old, mitochondria can turn into pro-death organelles, which produce reactive oxygen species that damage the cell’s proteins and DNA. This is a major problem for cardiac myocytes, which rely on the energy produced by mitochondria to contract. Additionally, the body cannot replace these particular cells because they do not divide.
Cells have various quality control mechanisms to detect and repair dysfunctional mitochondria, but when the organelles are too damaged, the cell degrades them using lysosomes. We wanted to determine what happens to the cell when the lysosomes are not functioning well or are overwhelmed, and if there was another pathway to temporarily deal with the damaged mitochondria. This information is of particular importance to patients with Danon’s disease, who have mutations in a lysosomal protein that causes cardiomyopathy.
How do cells dispose of damaged mitochondria without using their lysosomes?
We discovered that fibroblasts and cardiac myocytes secrete mitochondria inside extracellular vesicles (EV) when their lysosomal function is compromised or overwhelmed. This encapsulation ensures that the mitochondria do not elicit a dangerous immune response once outside the cell because of their bacterial origin. The mitochondria-containing EV originate from within multivesicular bodies (MVB), which either deliver the cargo to the lysosomes for degradation or ship everything to the plasma membrane for secretion. We found that Rab7, a protein present on the MVB’s outer membrane, is a regulator involved in dictating the fate of the vesicles. We believe that active Rab7 directs the EV toward the lysosomes, but in the absence of this protein or when it is inactive, the cell will traffic the EV to the plasma membrane.
Once cardiac myocytes release the mitochondria-containing EV, resident cardiac macrophages and other cells in the heart internalize the vesicles to degrade them through their lysosomes. The EV do not seem to enter circulation but stay within the heart. Ultimately, this is an alternative garbage disposal pathway used by cells to get rid of dysfunctional and damaged mitochondria when they cannot degrade the organelles in their own lysosomes.
Why did you use multiple microscopy techniques in this paper?
We used each microscopy technique to answer a different research question. We employed traditional fluorescence microscopy to look at the colocalization between the EV and mitochondria. We used spinning disk confocal microscopy for our live cell imaging because we needed a microscope that was fast enough to capture many mitochondrial sequestration events over a short period and was equipped with an environmental chamber to maintain carbon dioxide levels and temperature. Electron microscopy (EM) allowed us to look at the mitochondria and EV on an ultrastructural level, which is not possible with fluorescence microscopy. Using EM, we were able to assess the vesicle’s size and shape, as well as the mitochondrion’s morphology, including whether it was swollen or falling apart. CLEM, which I think is the best technique that scientists ever invented in terms of imaging, allowed us to confirm that mitochondrial fragments are inside the EV and not just nearby. CLEM ensured that what we considered colocalized using the fluorescent microscope was accurate.
What are your next steps?
In this study, we observed an increase in the secretion of mitochondria-containing EV after we stressed the cells. Additionally, cell death levels increased when we blocked secretion. The next step is to examine what happens in the diseased state. For instance, we want to determine if EV secretion will increase after we give the mice a myocardial infarction, which causes a great deal of mitochondrial damage. Furthermore, we want to investigate if the EV are contributing to cardiac remodeling and the activation of inflammation in the diseased state.
This interview has been condensed and edited for clarity.
Reference
1.Liang W, et al. Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nat Commun. 2023;14(1):5031.